Byline: Dr James Ellison
More than 12 million ambulance incidents were handled by UK ambulance services in 2017-18, a reminder that pre-hospital care is no longer a peripheral part of health systems. It is a strategic pressure point where time, data quality, and clinical judgement converge under severe operational constraints, as noted in CB Insights coverage of Remote Diagnostic Technologies. For G20 ministers, that makes remote diagnostic technologies ltd more than a company profile. It is a lens on how states can strengthen emergency response, extend specialist oversight into remote environments, and govern sensitive health data across borders.
Founded in 1997, Remote Diagnostic Technologies Ltd emerged from the UK’s pre-hospital emergency care ecosystem and was acquired by Royal Philips in June 2018, integrating its technology into Philips’ Therapeutic Care business in the €1.4 billion global resuscitation and emergency care market, according to CB Insights on RDT’s acquisition context. That story is usually told as a corporate transaction. The more important story is what happened to a niche UK emergency care capability once it became part of a multinational platform with reach across maritime operations, aviation, and wider digital health systems.
The policy question isn’t whether remote diagnostics matters. It already does. The question is whether governments will treat these capabilities as procurement line items, or as strategic infrastructure for health security, disaster response, and digital sovereignty.
Table of Contents
- The Strategic Importance of Remote Diagnostics
- Unpacking RDTs Tempus Platform Technology
- Notable Deployments and Performance Evidence
- The Philips Acquisition and Market Position
- Data Governance and Patient Privacy Implications
- Challenges and Future Outlook for Remote Diagnostics
- Actionable Recommendations for Policymakers
The Strategic Importance of Remote Diagnostics
Across the G20, the weak point in emergency care is often not clinical knowledge but the distance between the patient and the clinician who can act on it. Remote diagnostics addresses that gap by shifting care coordination from fixed facilities to connected networks, where monitored data, communications links, and decision support can extend specialist oversight into ambulances, ships, aircraft, and disaster zones.
RDT’s strategic significance should be read through that lens. Before Philips acquired the company in 2018, RDT had already built tools for pre-hospital and emergency use cases that linked monitoring, defibrillation, and data transmission in field settings. After the acquisition, those capabilities became part of a larger international emergency care portfolio. The policy implication is broader than one transaction. It shows how a niche pre-hospital technology can become national health security infrastructure once it is integrated into global supply chains, standards frameworks, and procurement systems.
For G20 governments, the strategic value lies in three areas:
- Operational reach: Remote diagnostics extends specialist input to frontline responders and isolated environments where treatment decisions must be made before hospital arrival.
- System resilience: Earlier transmission of clinical information can shorten handoff delays, improve triage, and preserve capacity during surges, disasters, or transport disruptions.
- Cross-sector utility: The same underlying capability supports civilian health systems, civil protection agencies, maritime and aviation safety, and humanitarian response operations.
These are not only technical gains. They change how states should plan emergency readiness. A connected diagnostic platform can serve as part of national response architecture, especially when ministries align health procurement with telecom resilience, cybersecurity rules, and interoperable data standards. That wider transition is reflected in current debates on supporting a shift from analogue to digital health systems.
The post-acquisition dimension is especially important. Philips did not merely buy a product line. It absorbed field-tested capabilities that could be deployed at greater scale across emergency and resuscitation workflows. For ministers, that is a signal about strategic dependency. If remote diagnostic capacity sits inside a small number of multinational platforms, public authorities need clearer rules on interoperability, vendor concentration, continuity of supply, and access during cross-border emergencies.
A second conclusion follows. Countries that want stronger emergency care systems should not treat remote diagnostics as a standalone device purchase. They should buy for integration, requiring compatibility with ambulance services, hospital information systems, public alert networks, and secure communications infrastructure. The states that do this well will be better placed to coordinate disaster response, support remote populations, and contribute assets to multinational health emergencies without rebuilding systems in the middle of a crisis.
Unpacking RDTs Tempus Platform Technology
Remote care systems succeed or fail on one operational question: can they move clinically useful information fast enough to change a decision before transport, deterioration, or costly evacuation occurs? Tempus was built for that narrow but consequential problem.
The platform combines physiological monitoring with voice and video transmission over cellular or satellite links. In practice, it allows a clinician at a distance to review a patient’s condition with more than a verbal handover, including readings such as ECG, SpO2, and NIBP. The same verified company record also notes reported reductions in response times in remote settings and fewer unnecessary evacuations in maritime use cases, according to the UK Companies House entry for RDT.
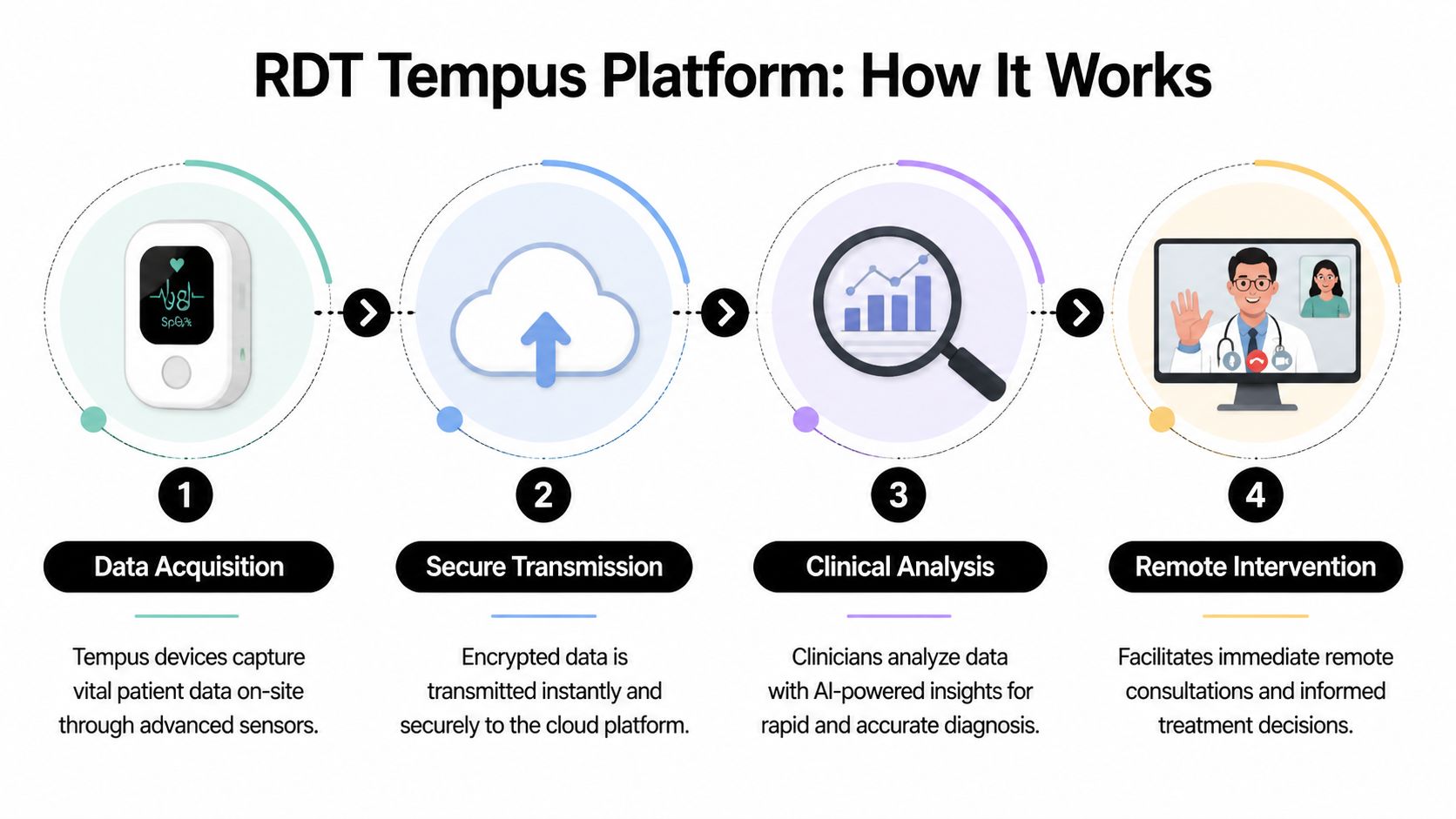
How the platform works in practice
Tempus operates as a connected clinical workflow rather than a single monitoring device. Its function is easiest to assess as a sequence of actions that compresses time between first contact and specialist input.
Capture at the scene
A responder records patient observations and monitored data at the point of care.Secure transmission
The system sends those data through available cellular or satellite networks to a remote clinical endpoint.Remote clinical review
A physician or receiving team assesses incoming information directly, instead of relying only on second-hand description.Directed intervention
The frontline team receives clearer guidance on treatment, transport priority, and whether escalation is warranted.
Remote medicine often breaks down when field staff can describe symptoms but cannot transmit enough evidence to support a confident decision. Tempus addresses that gap by combining monitoring, communications, and remote review in one chain.
Why the technology changes the decision cycle
The policy significance lies less in digitising observations than in changing where triage happens. Traditional emergency systems concentrate expertise at the receiving facility. Tempus shifts part of that expertise upstream, closer to the incident and earlier in the care pathway.
That has implications beyond operational convenience. In disaster zones, maritime incidents, austere military settings, and remote communities, the key constraint is often not the absence of a device. It is the delay between patient contact and authoritative clinical judgment. A platform that shortens that interval can improve asset allocation, reduce avoidable transfers, and preserve surge capacity for patients who need high-acuity transport.
Remote diagnostics works best when it supports a clinical chain of command, not when it acts as a standalone gadget.
For G20 ministers, the procurement lesson is clear. Governments should not buy connected devices in isolation. They should commission systems that fit national referral pathways, telecom resilience plans, cybersecurity requirements, and interoperable health data standards. That wider governance question sits within the broader debate about how digital health technologies are reshaping care delivery and public system design.
What Tempus represents beyond hardware
Tempus also illustrates why the post-acquisition phase matters for policy. Once a field-tested remote diagnostics capability sits inside a larger multinational platform, it becomes more than a product. It becomes part of national preparedness, cross-border medical assistance, and digital dependency management.
Historically, emergency care systems were designed around moving the patient to expertise. Platforms like Tempus begin to move part of the expertise to the patient before transport is complete. For health ministries, that shift changes how they should think about licensing, stockpiling, procurement, mutual aid agreements, and continuity of service during international emergencies.
Notable Deployments and Performance Evidence
A true test of remote diagnostics is whether it performs when isolation, time pressure, and operational cost all collide. That’s why RDT’s most telling deployments are not in low-stakes administrative settings, but in aviation and maritime operations, where a poor decision carries immediate safety and cost consequences.

Aviation as a policy proving ground
Post-acquisition, Tempus was deployed on over 3,500 aircraft, including UK-based carriers, and has been shown to reduce unscheduled landings for medical reasons by 40% across European fleets, with estimated operator savings of £2-5 million annually, while each diversion can cost an average of £50,000, according to 1TECHNATION’s report on the Philips acquisition of RDT.
For a policymaker, that evidence points to something larger than airline efficiency. It shows that remote diagnostics can alter decision thresholds in environments where evacuation is expensive, disruptive, and often avoidable if a clinician has better real-time information. Aviation is effectively a stress test for remote medicine governance. If a system can support safe clinical judgement at altitude, it can inform policy thinking for other difficult settings.
Maritime deployment and triage discipline
The maritime use case reveals a different policy benefit. Ships face distance, weather, and limited onboard capability. In those conditions, unnecessary evacuation isn’t just expensive. It also consumes scarce rescue assets and can distort regional emergency planning.
The evidence in the verified record indicates that Tempus deployments on ships operating in UK waters were associated with fewer unnecessary medical evacuations. That suggests a direct gain in triage discipline. In policy terms, triage discipline is one of the least discussed but most valuable outcomes of digital health infrastructure. It improves not only patient management, but also system-wide resource allocation.
A short visual summary is useful here:
What ministers should take from these deployments
These examples support three strategic conclusions:
| Setting | Policy relevance | Strategic lesson |
|---|---|---|
| Aircraft | Cross-border emergency protocols | Better onboard diagnostics can avoid unnecessary diversions while preserving safety |
| Ships | Maritime health resilience and rescue planning | Remote oversight can improve evacuation decisions and conserve response assets |
| Pre-hospital emergency care | National ambulance and emergency coordination | Data transmission matters most when it is embedded in clinical escalation pathways |
Operational test: A remote diagnostic system proves its value when it helps professionals decide not to escalate, as confidently as when it helps them escalate fast.
That’s the deeper significance of the performance evidence. The technology doesn’t just make emergency medicine more connected. It makes it more selective, and that selectivity is where a large share of the public value lies.
The Philips Acquisition and Market Position
In 2018, Philips moved to acquire RDT at a point when emergency care was becoming a contest over data flow, clinical coordination, and control of the first minutes of care. For G20 health ministers, that matters more than the transaction itself. The acquisition showed that remote diagnostics had shifted from a specialist product category into strategic health infrastructure.

Why Philips bought a strategic capability
Philips did not merely add another device business. It added an operational layer that links patient assessment, communications, and escalation across ambulance services, remote clinicians, and receiving hospitals. In practical terms, RDT strengthened Philips in a part of the care pathway where decisions are time-sensitive, fragmented across institutions, and often made before a patient reaches formal care.
That positioning has policy consequences. Health systems increasingly procure for continuity of decision-making, not only for hardware performance. A platform that can move clinical information from the field into command and treatment pathways gains influence over how emergency systems are organized, what standards they adopt, and which vendors become embedded in national response models.
This matters in crises.
Pandemics, mass-casualty incidents, maritime emergencies, and climate-related disasters all increase the premium on distributed clinical oversight. G20 governments therefore need to read the Philips-RDT deal as evidence that pre-hospital connectivity is becoming part of national preparedness capacity, alongside stockpiles, surge beds, and emergency logistics.
A market signal for industrial policy and resilience planning
RDT began as a UK specialist and ended up inside a multinational medtech group because its technology addressed a persistent system problem: how to transmit actionable patient information quickly enough to alter triage, transport, and treatment decisions. Small firms often produce this kind of capability. Large firms often control the procurement channels needed to scale it across borders.
That pattern creates a familiar policy trade-off. Acquisition can accelerate deployment, regulatory reach, and systems integration. It can also concentrate technical know-how, reduce visibility into product roadmaps, and shift a nationally developed capability into a governance structure shaped by global corporate priorities.
For ministers, the lesson is clear. Emergency medtech should sit within resilience policy, industrial policy, and digital policy at the same time. Governments that treat remote diagnostics only as a procurement line item will miss its wider function in health security and state capacity.
Three policy questions follow:
- Resilience policy: Should countries identify remote triage and field-to-hospital data transmission as priority capabilities in national preparedness plans?
- Industrial strategy: How will governments support domestic innovators long enough to scale, rather than losing strategic technologies before public systems capture their full value?
- Market governance: What reporting should be required after major medtech acquisitions to show whether promised gains in interoperability, deployment, and public benefit were delivered?
These questions connect directly to a wider debate about why health data governance is everyone’s business, especially when operational health technologies move from local innovation ecosystems into multinational platforms.
The post-acquisition implication for G20 governments
The main policy issue is no longer who bought RDT. It is who sets the rules for the infrastructure that RDT-type platforms now represent. If a small number of large vendors come to mediate remote assessment, emergency communications, and clinical escalation, procurement decisions will start to shape sovereign capacity in much the same way that telecoms and cloud contracting already do.
G20 governments should respond early. They can require interoperability testing in real service conditions, preserve competition through open standards, and link public procurement to audit requirements on security, uptime, and cross-border operability. They can also coordinate internationally on certification expectations for remote emergency platforms used in aviation, shipping, humanitarian response, and mutual aid deployments.
Viewed this way, the Philips acquisition was an early indicator of a larger shift. Remote diagnostics is becoming part of the institutional architecture of emergency care, and countries that govern it well will be better positioned to manage the next cross-border health shock.
Data Governance and Patient Privacy Implications
Remote diagnostics often gets framed as a connectivity challenge. That’s incomplete. It is also a governance challenge because it moves sensitive patient information across organisations, communications networks, and sometimes national jurisdictions in real time.

Privacy risk increases when care becomes mobile
A system like Tempus is valuable precisely because it transmits voice, video, and patient measurements from places where conventional infrastructure is weak. But that mobility creates policy questions that many procurement exercises address too late:
- Who controls the data when a patient is assessed in one jurisdiction and reviewed in another?
- Which rules apply if a platform is operated by a multinational vendor but used within a public health system?
- How should consent, auditability, and retention be handled during emergency use?
The verified record notes that the platform aligns with interoperability expectations in NHS Digital policy contexts and references GDPR compliance and standards-based integration in the broader Philips-RDT framing. For ministers, the lesson is that interoperability and privacy can’t be separated. A system that moves data efficiently but doesn’t clarify accountability creates operational risk, legal uncertainty, and public trust problems.
Interoperability is not neutrality
There is a common assumption in digital health policy that interoperability automatically solves governance concerns. It doesn’t. Interoperability means systems can exchange information. It doesn’t decide who may access it, where it may be stored, or how disputes are resolved if something goes wrong.
That’s why cross-border emergency diagnostics needs a governance model with at least four components:
| Governance issue | Why it matters in remote diagnostics |
|---|---|
| Data sovereignty | Emergency data may cross borders before national authorities can review handling rules |
| Clinical accountability | Remote advice can influence treatment decisions without a physician being physically present |
| Cybersecurity | Mobile transmission expands the attack surface beyond hospital networks |
| Record integration | Data must be usable inside national systems without creating fragmented patient records |
Health data governance becomes harder, not easier, when the most valuable data is generated outside hospital walls.
Wider policy thinking on health data as everyone’s business gains relevance. Ministers need frameworks that define trusted use across ministries, providers, private vendors, and emergency services.
The practical governance standard governments need
Procurement teams often focus on device certification and connectivity specifications. Those are necessary, but they aren’t enough. Governments should also require clear contractual terms on localisation, secondary use, breach notification, and integration with national health records. In emergency medicine, governance failures don’t usually appear first as legal abstractions. They appear as delays, missing records, and uncertainty about who is responsible for the final clinical call.
Challenges and Future Outlook for Remote Diagnostics
The promise of remote diagnostics is real, but post-acquisition scale isn’t automatic. In the RDT case, the gap between capability and documented public-system integration is now one of the most important policy questions.
A critical view appears in the verified material tied to Philips’ acquisition context. It notes UK MHRA adverse event reports in 2024-2025 showing 12 incidents for Philips pre-hospital monitors versus 8 for competitors, alongside the lack of specific mention of RDT in the UK government’s £50m paramedic tech investment, suggesting that the technology’s potential hasn’t translated into broad, clearly documented NHS integration, as reflected in the Philips acquisition materials referenced in the verified dataset.
Where scale becomes difficult
This matters for ministers because health technology adoption often stalls after acquisition for reasons that are organisational rather than technical. Several issues stand out.
- Integration complexity: Once a specialist platform enters a large multinational portfolio, local fit can become harder to demonstrate, especially in public systems that require procurement evidence and workflow adaptation.
- Evidence visibility: A technology can be capable and commercially deployed, yet still lack the public evidence base needed for national adoption decisions.
- Regulatory confidence: Adverse event reporting doesn’t prove systemic failure, but it does raise the threshold for transparent follow-up and quality assurance.
The policy gap is not only technical
There is also a strategic communications problem. If governments are investing in paramedic and emergency technology but cannot easily identify where a platform like Tempus fits into national pathways, the issue may be less about product quality than about institutional visibility. Innovation gets lost when procurement, clinical leadership, and industrial strategy operate on separate tracks.
Strategic warning: A strong device can disappear in policy terms if no one documents how it performs inside public systems.
What the future likely depends on
The future outlook for remote diagnostic technologies ltd, and similar platforms, will depend on whether health systems can move from isolated deployments to governed service models. That means defined clinical use cases, transparent incident reporting, and better post-market evidence within ambulance trusts, air ambulance services, maritime networks, and emergency transfer systems.
The central challenge isn’t whether remote diagnostics can work. The evidence already shows it can in specific contexts. The challenge is whether governments can convert promising tools into repeatable, accountable public infrastructure.
Actionable Recommendations for Policymakers
G20 ministers should treat the RDT case as a practical policy template. It shows that remote diagnostics can support emergency care in difficult environments, but it also shows that scale requires governance, evidence, and institutional design.
Build standards around emergency data pathways
Governments should move beyond generic interoperability language and define minimum operational standards for emergency remote diagnostics. Those standards should cover transmission security, minimum data fields for remote review, escalation protocols, and integration into national clinical records. The aim is to ensure that remote monitoring data isn’t merely visible, but actionable and auditable.
Fund pilots that answer system questions
Public funding should support deployment models that test more than technical performance. Pilots need to answer harder questions:
- Clinical workflow: Who receives the data first, and who is authorised to advise?
- Operational fit: Does the platform reduce unnecessary escalation without delaying necessary escalation?
- Governance readiness: Can the system fit domestic privacy, procurement, and liability rules?
Ministers should insist that publicly backed pilots produce publishable lessons for ambulance, maritime, aviation, and disaster-response settings.
Use procurement to create trusted public-private partnerships
The Philips-RDT case shows the value of pairing specialised innovation with global scale. Governments should use procurement to shape those partnerships, instead of just reacting. Contracts should require transparent outcome reporting, incident disclosure, and compatibility with public digital health architecture. That enables governments to secure both innovation and accountability.
Coordinate health security across sectors
Remote diagnostics is one of the few technologies that sits naturally across ministries. Health ministries need it for emergency care. Transport ministries need it for aviation and maritime safety. Interior and civil protection authorities need it for disaster response. G20 countries should create joint frameworks so these sectors don’t procure overlapping systems that cannot share data or support common response protocols.
Treat field diagnostics as strategic infrastructure
The final recommendation is the broadest. Governments should classify advanced pre-hospital diagnostic capability as part of national resilience infrastructure. That means linking industrial policy, emergency preparedness, and digital governance. The countries that do this well won’t only buy better devices. They’ll build faster, more coordinated decision systems.
Global Governance Media tracks the policy choices shaping health security, digital governance, and international cooperation. For ministers, advisers, and institutional leaders working on emergency care resilience, explore Global Governance Media for analysis that turns complex systems questions into practical policy action.